Tyler Ford
02/03/2022
Antibiotics have been an essential tool of modern medicine since the 1940s. They’ve cured countless bacterial infections and saved many lives. Unfortunately, many bacteria are evolving resistance to antibiotics. In addition, other microbes like fungi, viruses, and parasites are also evolving resistance to the drugs used to treat them. Researchers call this cumulative problem “antimicrobial resistance.” This growing threat results in more than 1,000,000 deaths per year globally. That number is expected to increase as the impacts of this “silent pandemic” become ever more prevalent.
This series aims to provide a finer point on the impacts of antimicrobial resistance. It highlights some of the antimicrobial resistant pathogens that the US Centers for Disease Control (CDC) considers “Urgent Threats.” These include:
- Neisseria gonorrhoeae
- Candida auris
- Clostridioides difficile
In this post we cover the drug-resistant fungus, Candida auris. This pathogen can be found infecting patients in long-term care facilities and hospitals. It is often deadly if it causes a bloodstream infection.
Check out our previous infectious disease spotlights here: influenza, HIV/AIDS, tuberculosis, and N. gonorrhoeae.

Candida auris general biology and transmission
Candida auris is a fungus that can live and reproduce both as single cells (yeast cells) and in biofilms – connected bunches of cells that are often resistant to disinfection and antimicrobials. Biofilms can also persist on a variety of surfaces.
A rising concern, C. auris can often be found on surfaces in healthcare facilities and patients can acquire C. auris infections in the hospital setting (“nosocomial infections”). C. auris can be transmitted between patients via physical contact or through contact with any contaminated surfaces. From September 1, 2020 – August 31, 2021 over 1,100 confirmed and probable clinical cases were reported to the CDC. Targeted screening identified over 3,000 additional patients colonized with C. auris (CDC website).
Importantly, a person can be colonized with C. auris without having an active infection. That is, a person may harbor the fungus on their skin without any health effects. However, this colonization can potentially lead to future dangerous bloodstream infections. Some common symptoms of bloodstream infections include fever and chills, but those who get bloodstream infections often have other underlying conditions that may have similar symptoms. Mortality estimates for C. auris bloodstream infections vary widely and require more study but are quite high (30 – 60%) (Lockhart et al 2017, Chen et al 2020).
Candida auris is similar to other fungal pathogens such as Candida albicans but is not as well studied because it is so new. Careful study is hindered by the fact that C. auris is difficult to identify and comes in a diverse set of strains with different characteristics. To get a fuller understanding of its biology, researchers must study representative examples of these various strains together. In addition, some strains are difficult to manipulate genetically. Nonetheless, genetic tools like CRISPR have been used successfully in this pathogen. Researchers are learning more about C. auris everyday (e.g. Ennis et al 2021).
The environmental reservoirs of C. auris and its origins are unknown, but this pathogen may persist on non-living surfaces in hospitals and other healthcare facilities (CDC website, Welsh et al 2017, Kumar et al 2019, Vallabhaneni et al 2016,). At-risk patient groups include the elderly, premature babies, immunocompromised individuals, people with serious underlying medical conditions and those who’ve used broad spectrum antibiotics, had intravenous catheters, stayed in long-term care facilities, and had extended stays in healthcare settings (Hu et al 2021, CDC website).
Candida auris diagnosis
Candida auris has very similar morphology to other fungal pathogens and is therefore difficult to detect by culture alone. That is, it can be difficult to identify C. auris by growing it on petri dishes and inspecting the dishes. As you can see in the photo from the CDC below, C. auris can take on a variety of colors when grown on standard plates used for pathogen identification:
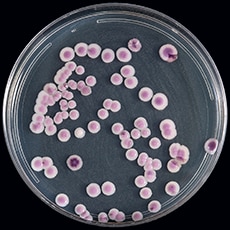
In fact, cases of C. auris infection going back to 1996 have been retrospectively identified using precise diagnostic techniques. PCR, DNA sequencing, or mass-spec based diagnostic techniques can be used to identify this pathogen but these require expensive, specialized equipment.
Candida auris treatment
There are a variety of antifungals available to treat C. auris. You can find an overview of antifungals and their mechanisms of action in this C. auris review. These kill fungi through mechanisms such as disrupting the cell wall or inhibiting RNA/DNA synthesis. Unfortunately, many strains of C. auris are resistant to one or more antifungals (Chow et al 2020) and this resistance may make C. auris a particularly problematic pathogen in the future.
Preventing Candida auris infection
With its high level of antifungal resistance, prevention measures are particularly important for controlling the impact of C. auris. The CDC recommends that patients colonized by C. auris be placed in single rooms to prevent additional spread of the pathogen. In addition, hospital surfaces must be disinfected with particular types of chemicals to effectively clear out any C. auris. On a personal level, individuals are encouraged to practice good hand hygiene with proper hand washing technique to prevent the spread of C. auris. Antifungal use is not recommended for patients who are colonized but not actively infected with C. auris.
Acknowledging the rising threat of C. auris, the WHO and the CDC are working to support and develop tracking infrastructure. With adequate surveillance, healthcare providers may be able to stop C. auris outbreaks before they result in high mortality.
Finally, a vaccine developed to treat Candida infections has been shown to protect mice (Singh et al 2019) from C. auris infection. A Phase Ia/IIb clinical trial using this vaccine to treat a different kind of fungal infection was completed in 2018 (Edwards et al 2018). More research is needed to determine if this or similar vaccines will be effective against C. auris.
Outlook
Fungi are often left out of the discussion of antimicrobial resistance, but antifungal resistance, like antibiotic resistance, is a growing issue. It’s important for public health organizations to expand their fungal monitoring efforts and it’s great to see that researchers are taking an interest in this area. With the growth of monitoring efforts, diagnostics (such as CRISPR diagnostics), and vaccines, we may be able to keep on top of the rising tide of antifungal resistance.